SAVR always better for younger AS? 'Durability data up to 10-years greenlights TAVR for patients between 65-70'
Long-term valve durability becomes crucial for younger AS populations; intermediate-term data strengthens case for TAVR in select patients under 70 years
"Meaningful" long-term data on valve durability are lacking, an expert said, but existing ones can aid decision-making between transcatheter aortic valve replacement (TAVR) and surgery in younger patients with aortic stenosis (AS) at lower operative risk.
 "Long-term valve durability has surfaced as the most important issue for decision-making between TAVR and surgical aortic valve replacement (SAVR), especially in younger patients with longer life expectancy and few comorbidities," said Duk-woo Park, MD, PhD (Asan Medical Center, Seoul, South Korea) at the Grand Walkerhill Seoul in South Korea during the AP VALVES & STRUCTURAL HEART 2022 conference on Aug 11.
"Long-term valve durability has surfaced as the most important issue for decision-making between TAVR and surgical aortic valve replacement (SAVR), especially in younger patients with longer life expectancy and few comorbidities," said Duk-woo Park, MD, PhD (Asan Medical Center, Seoul, South Korea) at the Grand Walkerhill Seoul in South Korea during the AP VALVES & STRUCTURAL HEART 2022 conference on Aug 11.
"What matters is durability beyond 10 years because more TAVR candidates are younger with fewer comorbidities," Park said. "But meaningful long-term durability data on TAVR valves are expected no sooner than 2025."
"Current evidence says TAVR can be considered for younger patients between 65 and 70, provided that coronary tomography angiography (CTA) shows anatomy suitable for TAVR with no other co-existing aortopathy, valve or coronary issues, and when patients are strongly opposed to surgery," he said. "Considering the increasing importance of lifetime management of AS, the ‘TAVR first' approach should ensure future coronary access, future TAVR-in-TAVR and future SAVR after TAVR."

Age becomes crucial for optimal decision-making between TAVR, SAVR
An optimal AVR strategy for younger patients (<75 years) at lower operative risk has become a point of contention among cardiologists, primarily due to bioprostheses' durability.
 Although SAVR and TAVR are the two major treatment methods for severe, symptomatic AS, SAVR had reigned supreme as the standard of treatment until the relatively novel, less invasive TAVR procedure emerged in 2002 with the first operation by Alain G. Cribier, MD(University of Rouen's Charles Nicolle Hospital, Paris, France)1.
Although SAVR and TAVR are the two major treatment methods for severe, symptomatic AS, SAVR had reigned supreme as the standard of treatment until the relatively novel, less invasive TAVR procedure emerged in 2002 with the first operation by Alain G. Cribier, MD(University of Rouen's Charles Nicolle Hospital, Paris, France)1.
The landmark PARTNER 3 trial2, published in the New England Journal of Medicine (NEJM) in May 2019, strengthened the case for TAVR, reporting significantly lower rates with TAVR than surgery for the composite of death, stroke or rehospitalization at 1-year, and no interactions between age and primary endpoints or signals of weaker durability with TAVR bioprosthesis compared to SAVRs.
The results, along with accumulated data favoring TAVR, led the U.S. Food and Drug Administration (FDA) to grant expanded approval for TAVR in younger patients at low surgical risk in 2019, shifting recommendations of major guidelines on both sides of the Atlantic.
In 2020, the American College of Cardiology and American Heart Association (ACC/AHA)3 guidelines strongly recommended TAVR (Class I) in patients over 65 - which was markedly bolder than the 2021 European Society of Cardiology and European Association for Cardio-Thoracic Surgeons (ESC/EACTS) guidelines' endorsement for TAVR (Class I) in patients over 75.
"The data supplements of the ACC/AHA guidelines4 recommended TAVR for older patients with fewer expected remaining years of life or patients with calcific AS and a tri-leaflet valve; concurrent cardiac conditions included severe calcification of the ascending ‘porcelain' aorta," Park said. "SAVR was deemed better for younger patients with longer life-expectancy, bicuspid aortic valves (BAVs) and subaortic (LV outflow tract) calcification, but carried risks of patient-prosthesis mismatch (PPM), which require consideration of annular enlargement."
TAVR applies to younger patients (65-70 years) who are strongly opposed to surgery if they have CTA-based anatomy suitable for TAVR with no other co-existing aortopathy, valve or coronary issues.
Despite the internationally strengthened endorsements for TAVR, the lack of reliable long-term data on valve durability confounded the issue of an optimal strategy for the patient population. The high risk of repeat procedures in younger patients further confined TAVR to the elderly population at high surgical risk.
"Currently, surgeons and interventionalists agree that TAVR is always better for patients over 80 years of age," Park said. "The decision becomes arguable and negotiable for patients between 70 and 80 years of age, requiring consideration of patient preferences."
"For patients under 70, surgeons and interventionalists strongly disagree on the optimal procedure with surgeons sticking to the concept of ‘SAVR is always better.'"
Cardiac surgeons have maintained the superiority of SAVR for the wide spectrum of AS patients, Park said, particularly owing to two major concerns of paravalvular (PVL) thrombosis and valve durability
PVL thrombosis not linked to strokes, neurological dysfunction: ADAPT-TAVR
Despite concerns about TAVR-related thrombosis and the consequent risk of stroke, findings from the ADAPT-TAVR trial5 showed that the "very common" incidence of PVL thrombosis was not causally associated with brain lesions or neurological dysfunction.
The study, also presented by Park at the American College of Cardiology 2022 (ACC 2022) conference and simultaneously published in the Circulation in April, found "very high rates" of PVL thrombosis were not significantly associated with new cerebral thromboembolic lesions or other neurological endpoints.
Analysis of PVL thrombosis between edoxaban and dual antiplatelet therapy (DAPT) arms showed no significant difference between leaflet thrombosis (9.8% vs. 18.4%), subvalvular thrombosis (27.5% vs. 26.6%), supravalvular thrombosis (0% vs. 1%), sinus of Valsalva thrombosis (12.8% vs. 22.0%) or any thrombosis at aortic valve complex dimension (37.3% vs. 48.6%).
As principal investigator, Park said: "For younger populations, the two major concerns of TAVR (and the underlying rationale for the ‘SAVR is always better' concept) include the risk of thrombosis and deteriorating valves but ADAPT-TAVR did not find any causal association between leaflet thrombosis and brain lesions or neurological dysfunction."
TAVR durability data up to 10 years - including the randomized NOTION, CoreValve US Pivotal and SURTAVI trials - shows no safety concerns compared to SAVR.
The problem of valve durability, which "absolutely" matters, was also not limited to TAVR, he added.
"Previous studies showed durability of bioprosthetic valves for SAVR were regarded to be 13 years at most; however, VIVID Registry findings on 1,304 aortic valves - presented at the TVT conference in 2016 - showed that the median duration of SAVR valves was 9 years, and more than half failed by 10 years."
The NOTION trial6, which examined structural valve deterioration (SVD) in TAVR and SAVR valves, also found significantly lower rates of SVD with TAVR at 8 years (SAVR 28.6% vs. TAVR 13.9%; p=0.0017).
5-year combined data analysis7 of the randomized CoreValve US Pivotal and SURTAVI trials - which examined SVD rates in transcatheter (n=1,128) and surgical aortic bioprostheses (n=971) - also found significantly lower SVD rates with TAVR compared to surgery at 5-years (SAVR 4.38% vs. TAVR 2.20%, p=0.004). Trends favoring TAVR were more prominent in smaller valves (<23 mm) and annular diameters (SAVR 5.84% vs. TAVR 1.32%, p=0.02).
Although the aggregate of these trials on valve durability showed no safety concerns with TAVR compared to SAVR, pursuing a strategy of "TAVR first" still requires caution, Park said.
"Younger patients may face up to 3 cycles of implants and explants, indicating that operators should pursue TAVR first only if it allows for future coronary access, future TAVR-in-TAVR and future SAVR after TAVR," he said. "We need more data on developing surgical techniques for TAVR explants, which are technically challenging due to issues like adherent stent frames in patients who are very sick at baseline."
Explant risks also extend to SAVR, Park said. A study published in the Annals of Thoracic Surgery8 on the long-term durability of bioprosthetic aortic valves in SAVR procedures (n=12,569) found a total of 354 surgical explants in younger patients at 10 years, caused primarily by SVD (44%) or endocarditis (41%). The 10-year mortality rate before explant was 46% and 76% at 20 years.
TAVR first' should be done if it allows for future coronary access, future TAVR-in-TAVR and future SAVR after TAVR.
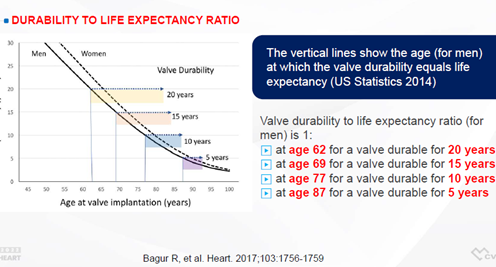
As a general rule, Park concluded that valves should match patient life expectancy, referencing the valve durability-to-life expectancy ratio9 outlined by Rodrigo Bagur, MD, PhD (Western University, Ontario, Canada) and colleagues in the BMJ Heart in 2017.
The 1:1 valve durability-to-life expectancy ratio for male patients suggests that valves should last 20 years for 62-year-old patients at the time of implantation, 15 years for 69-year-olds, 10 years for 77-year-olds and 5 years for patients over 87 years.
"Use of bioprostheses in younger patients has increased exponentially compared to mechanical valves based on the STS database10 and NVT database11," Park said. "But major valves, including mechanical and tissue valves, require weighing the risks and benefits for each patient."
"Although surgeons strongly advocate SAVR for patients between 50 and 69 years, 3 assumptions must be true to guarantee better outcomes with SAVR: surgical bioprostheses outperform transcatheter ones; sutureless bioprostheses12 outperform transcatheters’; and bioprosthetic materials for SAVR or TAVR are manufactured by the same company," he said.
"5- to 10-year durability data on TAVR, including the randomized NOTION, CoreValve US Pivotal and SURTAVI trials that used standardized definitions showed no safety concerns compared to SAVR in younger patients," Park added. "However, various strategies should be considered for younger patients undergoing TAVR, considering the importance of lifetime management of AS and the potential benefits of SAVR and the Ross procedure."
Edited by

Jung-Min Ahn , MD
Asan Medical Center, Korea (Republic of)
Written by

YoonJee Marian Chu, Medical Journalist
Read BiographyPark, Duk-Woo, et al. “Edoxaban versus Dual Antiplatelet Therapy for Leaflet Thrombosis and Cerebral Thromboembolism after TAVR: The Adapt-TAVR Randomized Clinical Trial.” Circulation, 2022, https://doi.org/10.1161/circulationaha.122.059512.

