CASE20230510_001
Repeat Transcatheter Aortic Valve Replacement to an Extremely Small Transcatheter Valve
By Juri Iwata, Yusuke Kobari, Kentaro Hayashida
Presenter
Juri Iwata
Authors
Juri Iwata1, Yusuke Kobari2, Kentaro Hayashida1
Affiliation
Keio University School of Medicine, Japan1, Rigshospitalet, Denmark2
TAVR - Valve-in-Valve TAVR
Repeat Transcatheter Aortic Valve Replacement to an Extremely Small Transcatheter Valve
Juri Iwata1, Yusuke Kobari2, Kentaro Hayashida1
Keio University School of Medicine, Japan1, Rigshospitalet, Denmark2
Clinical Information
Relevant Clinical History and Physical Exam
An 86-year-old woman had been experiencing chestpain and syncope during her dialysis sessions and she was diagnosed with severe aortic stenosis (AS) viatransthoracic echocardiography (TTE). She was noted to exhibit symptoms of New York Heart Association (NYHA)class 2. Her medical history included sick sinus syndrome. She receiveda permanent pacemaker. She had required hemodialysis for diabetic kidneydisease for 18 years. On admission, vital signs were stable at rest.
 Video 1.mp4
Video 1.mp4
Relevant Test Results Prior to Catheterization
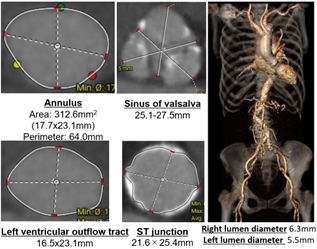
TTE showed a normal left ventricular ejectionfraction, an aortic valve area of 0.56 cm2, peak flow velocity of4.3 m/s, and mean pressure gradient of 41 mmHg. Brain natriuretic peptide (BNP)was 1,177 pg/ml. Computed tomography (CT) showed the aortic annulus to be 313 mm2in area and 64 mm in perimeter without significant calcification,the sinus of Valsalva to be 25-28 mm, sinotubular junction to be 22-25 mm. The Society of Thoracic Surgeonsscore for aortic valve replacement was 13.2 %.

Relevant Catheterization Findings
Our heart teamdecided to perform transcatheter aortic valve replacement (TAVR) with SAPIEN320 mm valve (Edwards Lifesciences) for the small annulus. The procedure wasinitiated with the right femoral artery as the main puncture site. A pigtailcatheter was placed through the contralateral left femoral artery and atemporary pacemaker was placed through the left femoral vein. The aortographywas performed in perpendicular view (left anterior oblique 15 caudal 5) for theplacement of the valve.
Interventional Management
Procedural Step
After deployment of the valve under rapid pacing,there was a residual moderate paravalvular leak with TTE. Immediatelyafter post-dilatation using the delivery balloon for four times, TTE revealedmoderate to severe transvalvular aortic regurgitation (AR). After conversion togeneral anesthesia, transesophageal echocardiography (TEE) revealed the stuck non-coronarycusp (NCC) leaflet, causing eccentric transvalvular AR. We decided to implantthe second valve (20 mm SAPIEN3) in the first valve because attemptsto fix the leaflet by a pigtail catheter was ineffective. Afterthe implantation of the second valve, TEE showed no transvalvular leak and thePVL of the first valve was decreased to mild. We also confirmed no significanttransvalvular gradient by invasive hemodynamic assessment using 2 pigtailcatheters (peak to peak pressure gradient of 5 mmHg). However, TTE one day afterthe procedure showed meanpressure gradient of 14 mmHg and a severeprostheses-patient mismatch (PPM) with an index effective orifice area (iEOA)of 0.50 cm2/m2. The patient was discharged home in goodcondition on the seventhpostoperative days after careful follow-up. Three weeks afterTAVR, her NYHA class reduced from 3 to 1, and BNP levels decreased from 1,177to 787 pg/ml, without any sequelae. The valves werewell expanded to almost circular shape with the internal diameter of 18 x 19 mmand an area of 264 cm2 observed on follow-up CT one month post-operatively.
 Video 4.mp4
Video 4.mp4
 Video 2.mp4
Video 2.mp4
 Video 3.mp4
Video 3.mp4
Case Summary
This report demonstrated a repeat implantation of the smallest transcatheter aortic valve(TAV). We noted that TTE and catheter assessment gave ‘discordance’ and marked improvement inclinical outcomes after TAVR showed theaccuracy and importance of invasive assessment. The incidence of PPM in Asiancohort was low due to smaller body surface area (BSA) than that of Westernpatients. BSA should be considered when selecting treatment options, and the 20 mm in 20 mm SAPIEN3 TAV-in-TAV should not be excluded in patients with smallBSA. As TAVR becomes more common in younger patients, the TAV-in-TAVprocedure may be a feasible option for patients with an extremely small annulusand small BSA.
