CASE20230511_001
A Immediately Post-implantation Severe Intravalvular Regurgitation: Engineering a Leaflet Issue?
By Wongsaput Boonyakiatwattana, Anuruck Jeamanukoolkit, Prayuth Rasmeehirun, Worawut Tassanawiwat, Tanyarat Aramsareewong, Piyanart Preeyanont
Presenter
Wongsaput Boonyakiatwattana
Authors
Wongsaput Boonyakiatwattana1, Anuruck Jeamanukoolkit1, Prayuth Rasmeehirun1, Worawut Tassanawiwat2, Tanyarat Aramsareewong3, Piyanart Preeyanont4
Affiliation
Chulabhorn Hospital, Thailand1, Sunpasitthiprasong, Thailand2, Phramongkutklao Hospital, Thailand3, Cardiac Center, Thailand4
TAVR - Complex TAVR
A Immediately Post-implantation Severe Intravalvular Regurgitation: Engineering a Leaflet Issue?
Wongsaput Boonyakiatwattana1, Anuruck Jeamanukoolkit1, Prayuth Rasmeehirun1, Worawut Tassanawiwat2, Tanyarat Aramsareewong3, Piyanart Preeyanont4
Chulabhorn Hospital, Thailand1, Sunpasitthiprasong, Thailand2, Phramongkutklao Hospital, Thailand3, Cardiac Center, Thailand4
Clinical Information
Relevant Clinical History and Physical Exam
An 83-year-old woman with severe symptomatic aortic stenosis (AS) was admitted with heart failure. She had hypertension, dyslipidemia and post ablation for supraventricular tachycardia. Echocardiography demonstrated tricuspid severe AS, a mean aortic gradient of 42 mm Hg, valve area of 0.78 sqcm, mild AR, and left-ventricular (LV) ejection fraction of 62%. The predicted mortality calculated by the STS was 5.12%. Given the intermediate surgical risk, the patient was considered for transfemoral TAVI.
 Video 1A.mp4
Video 1A.mp4
 Video 1B.mp4
Video 1B.mp4
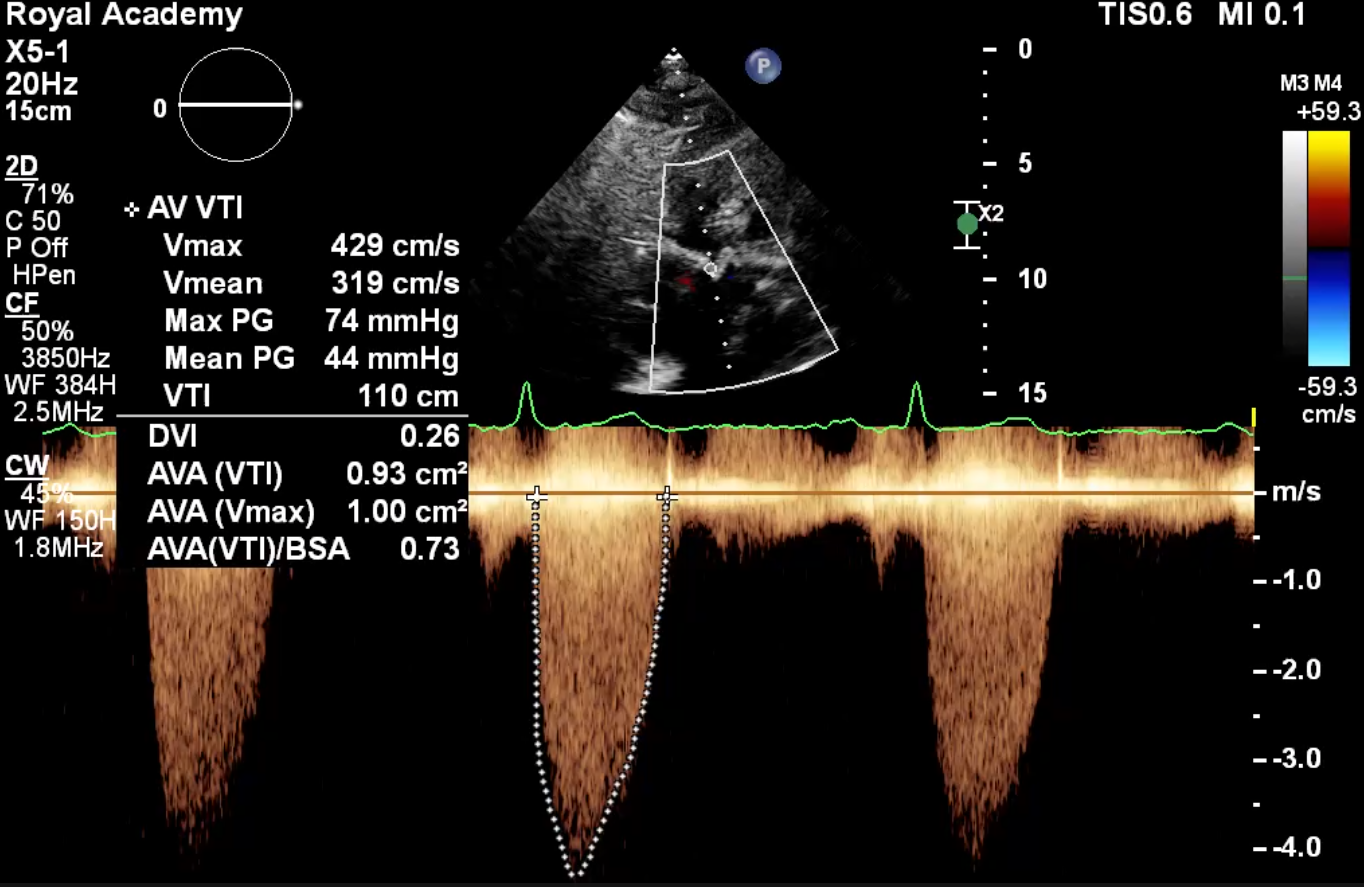

Relevant Test Results Prior to Catheterization
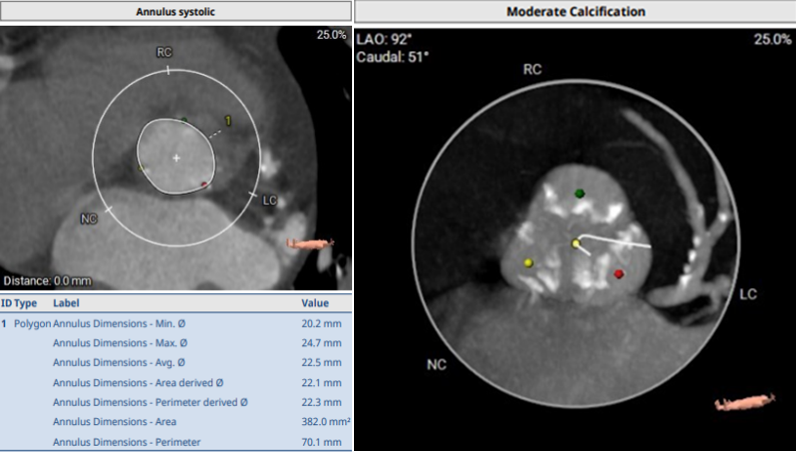
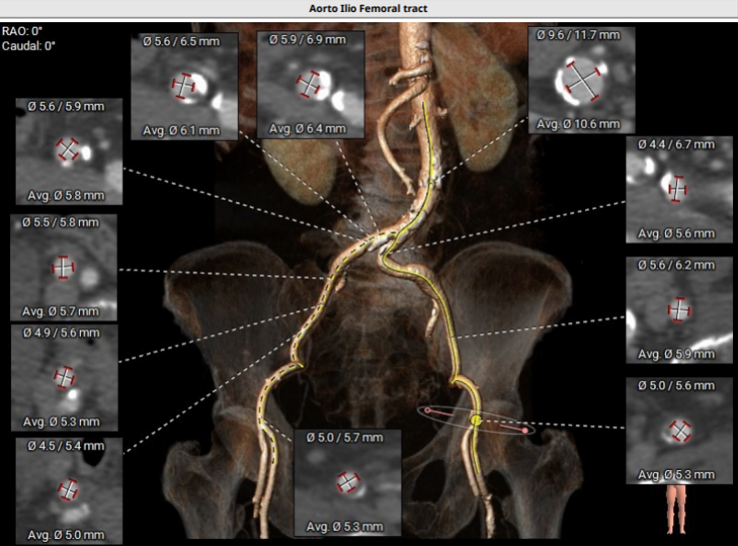
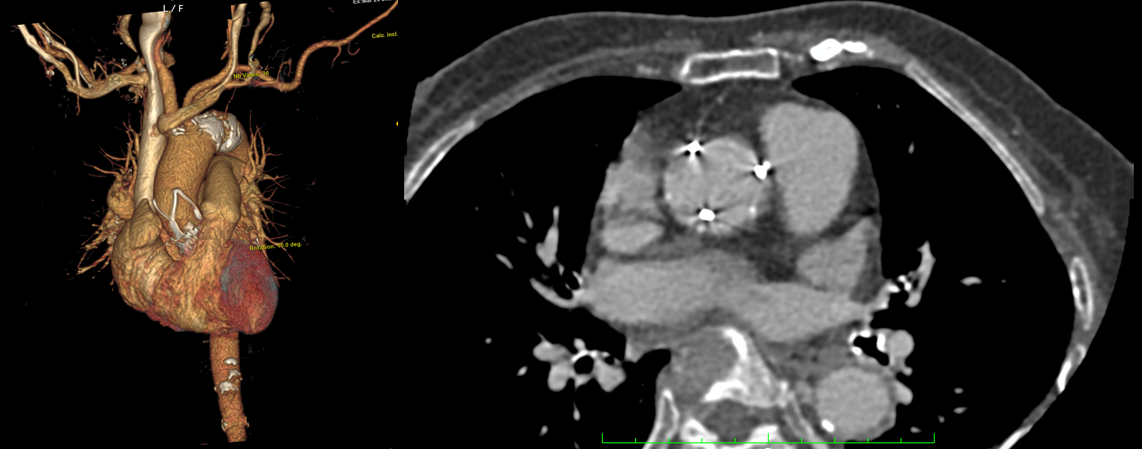
ECG revealed LVH with strain pattern. CAG revealed insignificant CAD. Pre-TAVI CT presented; moderately calcified symmetrical tricuspid valve with annular perimeter of 70.1 mm, derived-diameter 22.3 mm (Fig. 2A), average diameter of sinuses of valsalva (SOV) 29.9 mm, coronary ostia height 12.5 mm (left) & 13.4 mm (right), minimum femoral diameter 5.3 mm (right), 5.3 mm (left) and high bifurcation on both femorals (Fig. 2B). TAVI was planned under general anesthesia with Accurate Neo2 size S.


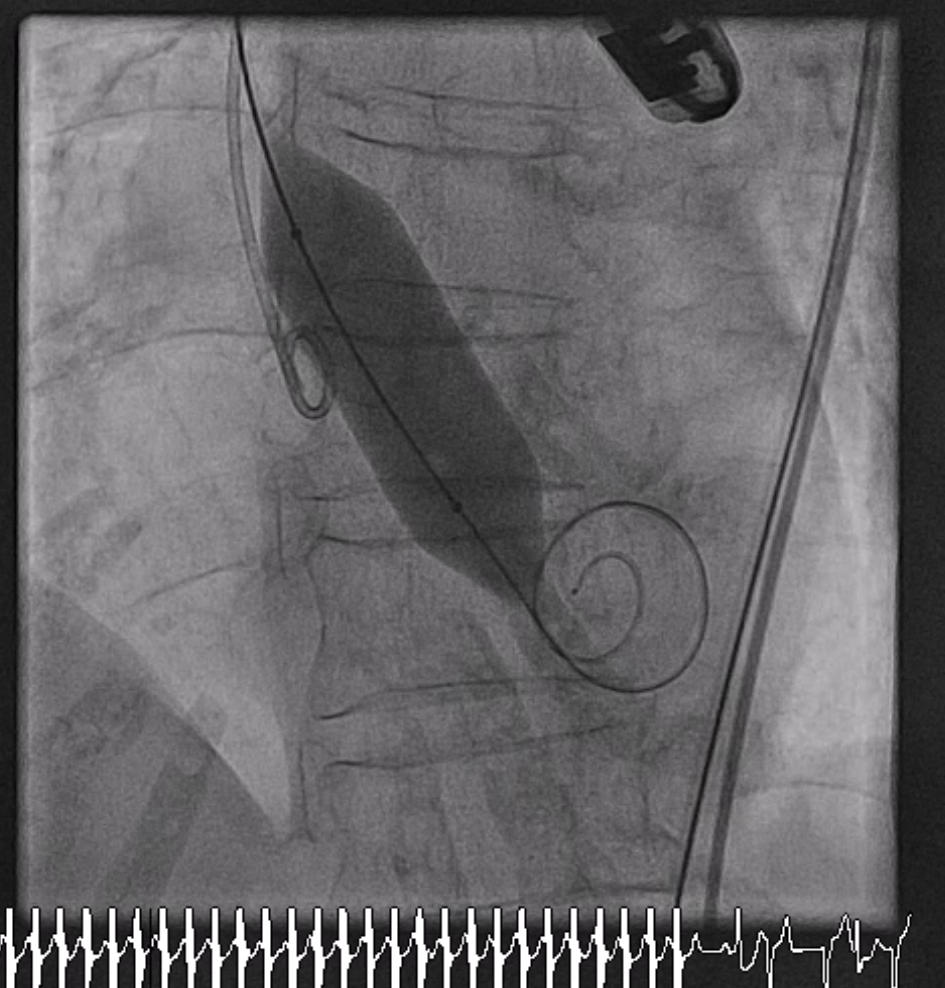
Relevant Catheterization Findings
The predilatation was done with 20 mmHg Numed balloon under rapid ventricular pacing (Fig. 3A). After predilatation of the aortic valve. TEE revealed mild AR. The Accurate Neo2 size S valve was adjusted for commissural alignment and implanted under TEE and angiography. The position of the valve was a little bit high but it’s still acceptable (Video 3B). However, a severe intravalvular leak was observed on TEE (Video. 3C) associated with high rising of LVEDP and LV dilatation.
 Video 3B.mp4
Video 3B.mp4
 Video 3C.mp4
Video 3C.mp4

Interventional Management
Procedural Step
Temporary intravalvular regurgitation during the procedure is commonly observed due to pressure of the stiff guidewire or catheter on the cusps inducing valve incompetence. However, this patient had persistent of severe AR after multiple attempts to reposition the cusp with the catheter and retrieval of the guidewire (Video 4A). In concern of severe AR, waiting for definite treatment, diastolic filling time was reduced by inotrope and high pacing rate. The mechanisms of intravalvular leak such as stent distortion, low implantation, incomplete deployment, and presence of a highly calcified aortic valve must be considered. TEE revealed an acceptable valve position and valve circular geometric shape with an immobile left coronary cusp. The mechanism is still unclear. We suspected that the valve’s immobility occurred from intrinsic tissue leaflet during crimping process. We decided to implant a second valve using the 23-mm Edwards–SAPIEN 3 (Video 4B). Angiography and TEE study showed complete resolution of the AR, and no paravalvular leak (Video 4C). Post-TAVI CT presented the 23-mm Edwards–SAPIEN 3 was placed in good position and the left cusp of Accurate Neo2 was immobile (Fig 4D). The crimping process was reviewed after end of the case but showed flawless. Nevertheless, there was a recent case report of mild intravalvular leak using with the same Lot of Acurate Neo2 Size S. So we alert the company to analyze about engineering leaflet issue.
 Video 4A.mp4
Video 4A.mp4
 Video 4B.mp4
Video 4B.mp4
 Video 4C.mp4
Video 4C.mp4

Case Summary
We describe a case of severe intravalvular leak due to an immobile cusp. Post-deployment leaflet injury accelerated the need for investigating to seek intrinsic leaflets problem. We need to consider the device issue not only complications in straightforward procedure but also repeating fault in same lot of device.The Acurate Neo2 size S in this lot was sent to analysis for hydrodynamics, leaflet mechanics, calcification susceptibility, crimping and deployment stability. Although this lot had a new manual suture design with qualified by in vitro ISO guideline, this design, specificly size S, may increased leaflet tension caused incompletely mechanical valve closing.
